Quality Business Leadership (QBL) repositions the Quality Organization as a Strategic Business Partner that focuses on optimizing and innovating in the areas of robust financial performance, sustained product quality, and consistent product supply (see Figure 1). This significantly differs from the Traditional Quality Support Function that primarily ensures cGMP compliance.
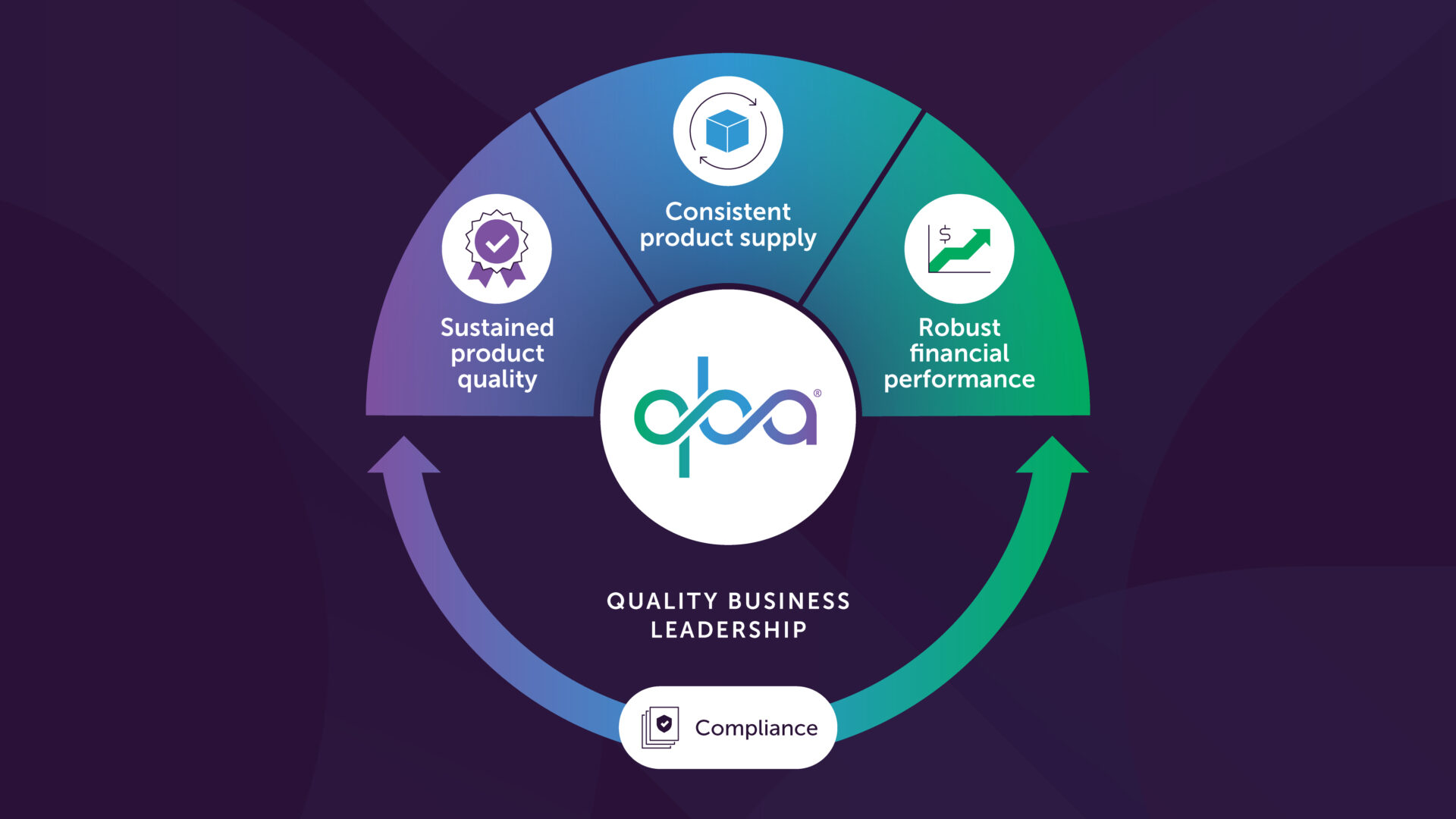
The Quality Business Leadership (QBL) Objectives
For the organization, the transition to QBL represents a widespread and fundamental shift in mindset on how the Quality Organization is perceived – moving from being seen as a reactive, necessary cost to an innovative, competitive, and value-driven asset.
Forward thinking leaders in organizations are now recognizing the value of integrating Quality as a Strategic Business Partner to the Executive Management and the Pharmaceutical Quality System (PQS) into the overall company business processes. They understand the necessity for Quality to be a priority owned across the organization, not just in one department, ensuring full alignment and support for success. They also realize the opportunity to differentiate themselves in the market as a trusted leader with fully optimized, highly efficient and effective systems that seamlessly serve patients in today’s uncertain world. In these organizations, the PQS is continuously simplified, accommodates to changes in supply, demand, business environment, and the implementation of new technologies.
For Quality professionals, transitioning to QBL requires unlearning inefficient and ineffective practices and mindsets while developing new skills and strategies. Quality Business Leaders master Six Key Traits and three QBL paradigm shifts (see Figure 2)
Six Key Traits
- Visibly demonstrate responsibility for quality
- Advocate that quality is owned by all through developing senior management partnership
- Engage employees to continuously improve quality
- Make risk-informed decisions that benefit patients
- Promote quality as a value-driven financial advantage
- Lead the path to one global quality regulatory framework
Three QBL Paradigms
- Focus on patients
- Lead more
- Think in systems
Traits and Paradigms of QBL
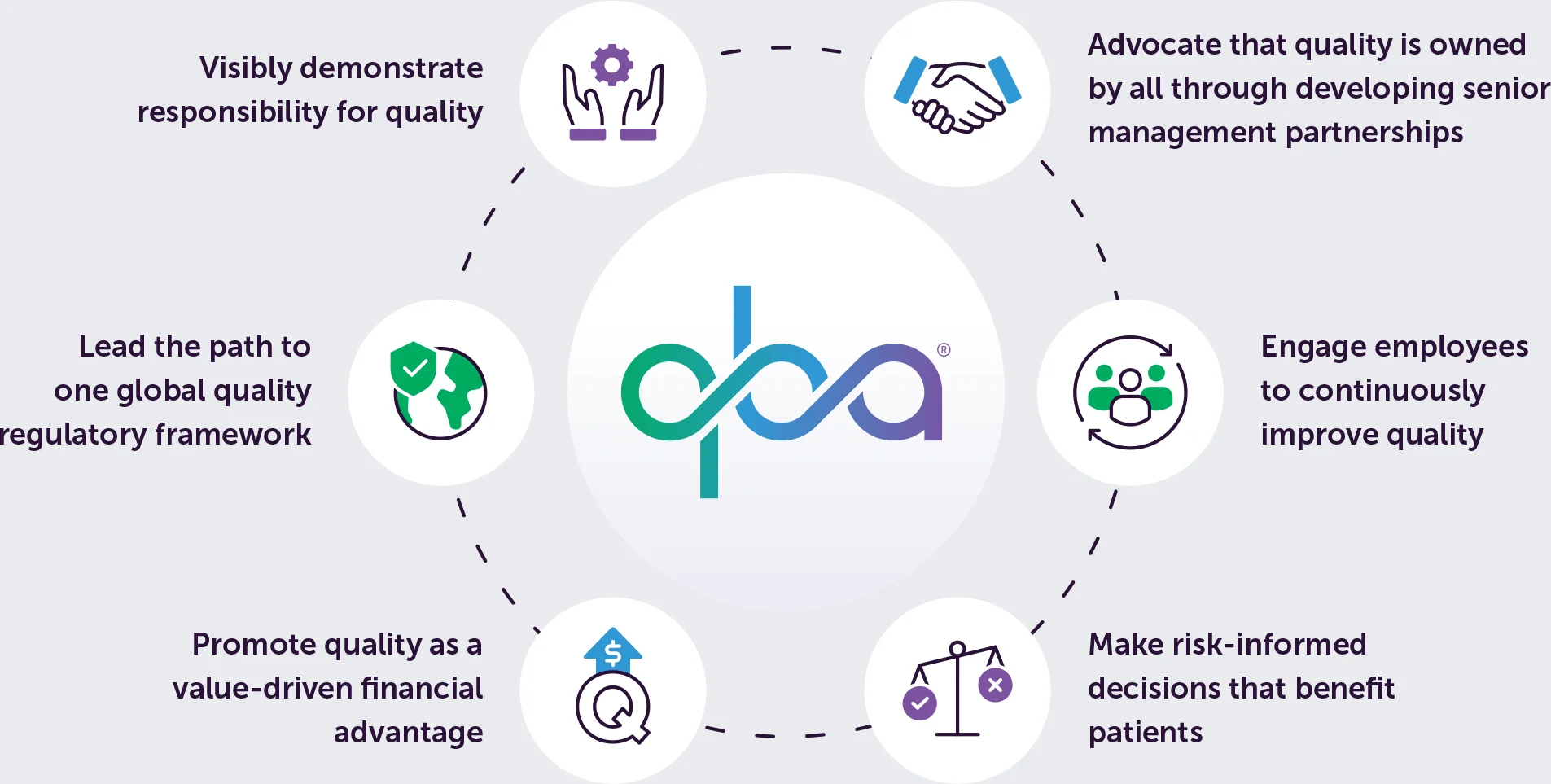
Trait: Visibly demonstrate responsibility for quality
Trait: Advocate that quality is owned by all through developing senior management partnership
Trait: Engage employees to continuously improve quality
Trait: Make risk-informed decisions that benefit patients
Trait: Promote quality as a value-driven financial advantage
Trait: Lead the path to one global quality regulatory framework
The Path from Traditional Support Function to Strategic Business Partner
Despite a stall in widespread action, senior executives have echoed Dr. Janet Woodcock’s aspirations for the future of Quality for decades. In 2004 she outlined FDA’s vision as “A flexible, agile pharmaceutical manufacturing sector that reliably produces high-quality drugs without extensive regulatory oversight.”
To achieve this vision, key changes must happen and the blueprint to get there is all within the Six Key Traits of QBL. QBL is a major catalyst for the necessary mindshifts and evolving processes and will prepare leaders and organizations for successfully navigating a complex and ever changing industry. If done effectively and collectively, organizations will recognize Quality professionals as key contributors within the corporate structure. Quality professionals will have also transitioned from a traditional compliance focused mindset (minimum expectations), to truly becoming Quality Business Leaders.
To kickstart the movement for change in our industry, 100 senior academia, regulatory agency and industry leaders met in May 2024 to develop a Quality Business Leadership Manifesto.
Quality Business Leadership Manifesto
Guiding the Next Generation of Quality Business Leaders
We believe quality is at the core of modern healthcare, serving to protect patients, strengthen organizations, and build the trust needed to sustain the industry. However, Quality leadership is at a turning point. Existing models won’t adequately meet the healthcare demands when technology is changing at a staggering pace and resources are more constrained. The opportunity now is for Quality Business Leaders to lead a transformation that demonstrates quality as financial value ensuring consistent product quality, strengthened supply, and sustainable long-term organizational success.
A New Standard for Quality Leadership
Simply meeting cGMP regulations is not sufficient for businesses to succeed and thrive. Real progress can begin when Quality Business Leaders are embraced as strategic partners who shape business outcomes, anchor decisions in quality, trust, and patient safety, and communicate quality with financial fluency.
Our work focuses on creating a practical mindset shift that moves Quality Business Leaders from support function to strategic business partner and a trusted driver of value. We go beyond traditional perceptions of quality through visible actions and measurable business outcomes.
The Quality Business Leader
This moment demands a new kind of leader: the Quality Business Leader. They champion:
-
Patient-centered decision-making, under uncertainty prioritizing safety, efficacy, and high-quality products while balancing science and compliance through rigorous risk-benefit thinking
-
Financial value through quality, demonstrating that strong quality leadership drives waste reduction, sustainable performance, and long-term success
-
Ownership and partnership, making quality a shared responsibility and mindset across the organization at all levels
-
Regulatory collaboration, contributing to a science-based, forward-looking global framework that supports continuous improvement, innovation, and global harmonization
-
Simplified, integrated quality system, integrated quality system, improving agility, effectiveness, supply reliability, robustness and resilience.
These are not abstract ideals. They are practical mindsets and behaviors that reshape organizations, elevate careers, and enable lasting business results.
The Future We Are Building
We envision a future where quality is fully owned by every employee and integrated into the business. Where risk is understood earlier. Where decisions move faster and with greater confidence. Where trust is strengthened across regulators, companies, and patients alike.
This future is already taking shape as quality leaders sharpen financial fluency, simplify systems, and expand their influence. Momentum is building.
A Call to Lead
If you have ever felt that your expertise was undervalued, believed Quality could contribute more than your role allowed, or wanted to lead but lacked the language to influence at the highest levels, this manifesto is written for you.
This is the moment for Quality Business Leaders to step forward with clarity, partnership, and confidence. This is our movement. And this is your invitation. Join the many Quality Business Leaders who are redefining what quality can achieve.
The QBL Imperatives: A Call to Action, The Change
The Quality Business Leadership Manifesto is a powerful call to action for senior leadership across Quality, Operations, and Regulatory Agencies.
The Manifesto signifies a crucial turning point. The change requires a united front, driven by both visionary leadership and a strong commitment from all levels within organizations. Regulators have a vital role to play in fostering this transition by adapting their approach to incentivize and support QBL implementation. Ultimately, the QBL will benefit all key stakeholders, garnering widespread societal support.
The goal is audacious: transforming the entire pharmaceutical industry from Traditional Quality Support Function to Quality Business Partner through the implementation of Quality Business Leadership (QBL).
Implementing these first steps is a good start on the journey to QBL:
- Become a QBL Advocate: Publicly endorse the QBL Manifesto and share it within and outside your company. This sends a powerful message to your company and the industry.
- Invest in QBL education: Allocate resources for QBL initiatives. This could include employee training in QBL principles or implementing new quality systems and processes.
- Start developing and implementing a Quality Value Index (QVI) model to track costs that are revenue, cost of goods and labor impacting respectively. This will allow you to identify improvement opportunities.
- Add Financial Risk Numbers (FRNs) to your risk register. FRNs are calculated as severity x occurrence x cost of correction / cost of prevention. FRNs add a risk to business dimension to your already existing risk to patient risk ranking.
- Encourage and support volunteers to initiate quality improvement projects and celebrate successes.
- Have leaders outside Quality communicate progress on quality goals and metrics. This will help the process towards quality owned by all.
Learn more about QBL and QBL Education here or by emailing Anders Vinther at anders@QBAleaders.com or by using our contact form.
Note: Quality Business Administration LLC (QBA) is a company dedicated to the education of Quality Business Leaders and support of companies transforming Quality to become Strategic Business Partners. QBA services include the Certificate in Quality Business Leadership Program accredited by the Technological University of Dublin. The Program is offered under the Graduate Business School as part of the MBA program.


