The two pharmaceutical industries
The pharmaceutical industry consists of two fundamentally different segments that are often treated the same – branded and generics. Taking a ‘one size fits all’ approach has proven ineffective in reducing drug shortages and improving quality performance. The drug shortage root cause analysis needs to get more granular so that ‘fit-for-purpose’ approaches can be applied to different companies. This article discusses how Quality leaders and senior-level regulators can help achieve a better state of drug product supply and quality management.
The generics drug industry operates with very low margins, which makes it less attractive for companies to enter or stay in the market. Today the average price for a 30-count top generic drug is significantly less than the cost of a cup of coffee at your local Starbucks, and generics pricing has continued to drop over the past years. The branded drug industry generally operates with higher margins but is faced with increasing price pressure, which means manufacturing cost-cutting is also expected in this segment over the next years.
Companies can only stay in business if they are profitable. If the cost of generic drugs keeps falling, we will continue to see most manufacturing in lower cost areas like India and China, and likely very few new companies will enter the market. Regulators and Quality leaders have no influence on the generics financial model (only politicians do), but we can articulate the consequences to quality and compliance problems when prices are as low as they are with the expectation to drop even further.
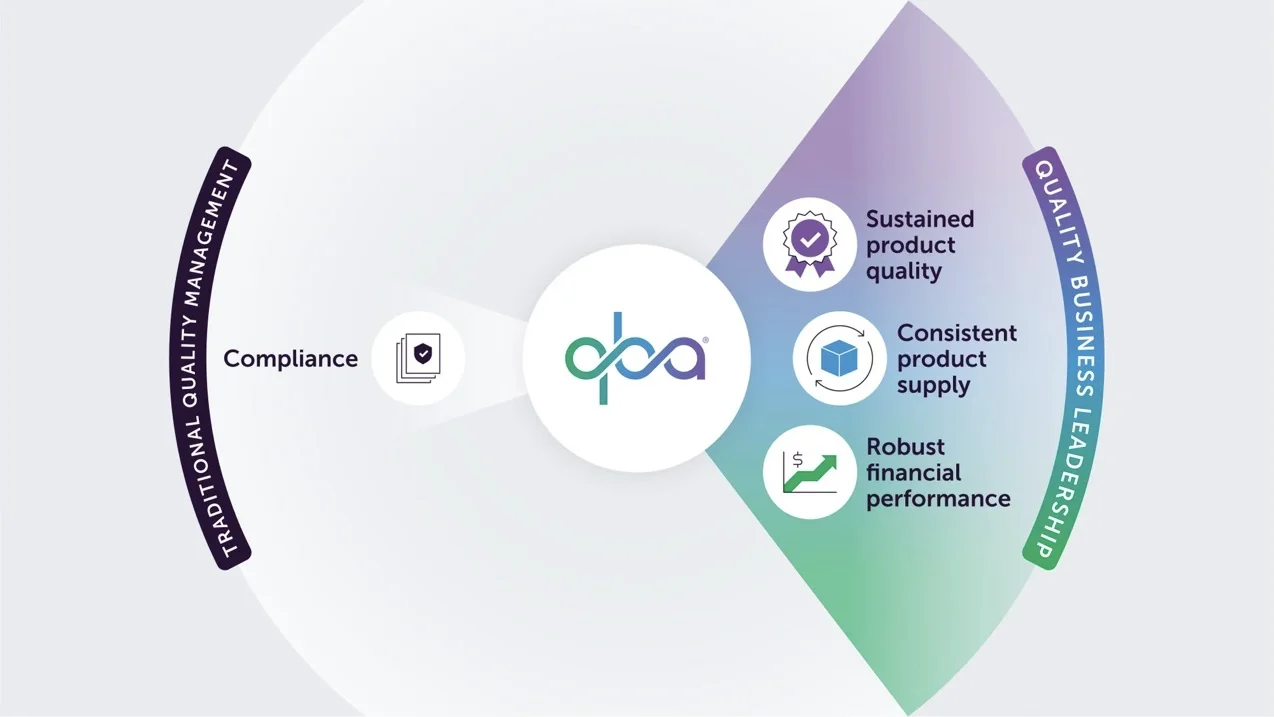
No two companies are the same
FDA and other regulatory agencies assess companies in various ways including inspections of manufacturing sites. If the company is significantly deficient in cGMP compliance it can lead to regulatory actions. In the USA this could be import alert, Warning Letter (WL) or other. When a WL is issued it can sometimes cause drug shortages if the manufacturer halts operations to fix the non-compliance or if an import alert is issued and no other manufacturer can promptly pick up the supply. A search was conducted for WLs issued over the past 5 years (from 2018 until 2023) in the Redica database for the top 24 branded drug companies (based on 2022 revenues) and the top 24 generics companies (based on most units dispensed per METYS/Symphony US market prescription data). The search revealed 3 WLs for 2 of the branded companies and 15 WLs for 9 of the generics companies. 14 of the 18 WLs (78%) were given to manufacturers for GMP violations at facilities in India.
Although there was a higher percentage of WLs issued to generics (38%) than branded companies (8%), it is important to note that within the two segments there are companies that demonstrate better quality performance than others. This should be reflected in how regulatory agencies manage individual companies with respect to reporting requirements. Companies that do not meet cGMP compliance requirements should be subject to additional controls, while those showing good quality performance should be treated with more trust. Today this is not the case. All companies are held to the same reporting requirements regardless of their quality performance. ICH Q10 (2008) and ICH Q12 (2019) allows for a gradation in that companies demonstrating an effective PQS and a good understanding of their processes and products, should be given regulatory flexibility whereby more post approval changes (PACs) can be handled in the PQS only or as a notification, rather than being submitted for prior approval. That flexibility, however, has not materialized. Instead reporting requirements have continued to increase for all companies over time.
More control and more trust
Reading the 18 WLs mentioned above was concerning as they showed non-compliance with basic GMPs. Examples are:
- Inadequate cleaning procedures causing contamination from one product into the next.
- Cross contamination from inadequately purified recovered solvents.
- Manual integration of chromatographic peaks to make OOS results pass.
- OOS results invalidated without reason or inadequately investigated.
- Facility or equipment not fit for purpose (water leaks, rust, inadequate practices).
- Selective release of batches despite failing process validation.
- Inadequate investigations (not finding root cause, not extending to other batches).
- Deadlegs in the water system causing microbial contamination.
Such non-compliance simply should not happen, and the consequences should be significant. In my view CEOs and CQOs of such companies should risk the ability to work in the pharma industry for a certain period. Controls should be increased at the company’s expense. This could include regulatory agency testing of batches prior to release to the market.
On the other hand, companies and manufacturing sites with good quality performance should experience less extensive regulatory oversight thus facilitating timely continual improvement of their operations and the Quality System. As an example, receiving approval of a PAC that requires regulatory agency prior approval from dozens of countries where the product is marketed, can take 3-5 years at a global scale (from first submission to last approval). Companies that demonstrate good quality performance should be trusted more and allowed the flexibility to manage most changes in the company’s Quality System only (rather than requiring regulatory agency prior approval) – as intended in ICH Q10.
Varying the level of regulatory oversight based on quality performance will incentivize all companies to do better. It will facilitate continual improvement for well-performing companies and make it costly for companies that do not meet basic GMP requirements.
From national to global approaches
We need to take a closer look at what the specific shortages are (generics or branded, type of manufacturing technology, geographic location of manufacturing, etc.) to be more successful in reducing them. It will provide a much better picture of where the issues exactly are e.g., it is my understanding that the majority of drug shortages are generic drugs. Then a root cause analysis should be performed to find out the reason for individual shortages. FDA states that a majority of drug shortages are caused by manufacturing quality problems. Assuming this is the case, what is the reason? Is it caused by the generics low-cost financial model, by the global regulatory complexity for PACs, by inadequate quality system for some companies, or other? A 5 Why analysis across different shortages, would most likely reveal there are several root causes, each requiring its own set of actions.
So far political and regulatory agency response to the drug shortage problem has been at a national level and with additional reporting requirements (and thus cost burden) for companies. Additional reporting has not translated into reduced drug shortages. Politically there is no coordinated effort at a global level to address drug shortages. Regulatory agencies have shown some collaboration across countries, but guidance documents are still at a national level like the FDA Quality Management Maturity (QMM) program. Reduction of the global problem of drug shortages requires a global approach and coordination.
One group of experts has been overlooked in the drug shortage discussion and yet they are directly responsible for release of drug products to patients: the Quality Assurance (QA) function of pharmaceutical companies. QA manages and navigates through the many national requirements when they release a batch of drug product to patients. They should speak up more. They should be asked and included in the dialog more.
The voice of Quality leaders
I’ve been a senior Quality leader for many years and have first-hand experience navigating the many different national regulatory frameworks to both stay cGMP compliant and ensure uninterrupted supply of drug products and vaccine to people. It’s not easy.
Industry Quality leaders have no decision right when it comes to regulatory requirements, which are set at a national level by the regulators. However, we must comply with them all. I’ve had the task of merging more than 10 different change management systems across a company. Experts were passionately debating if there should be two, three, or four levels of changes. The fact is, it doesn’t really matter, as long as you apply sound science, are data-driven, and meet GMP requirements. I had the privilege of being the final decision maker and we developed one change management system (IT, business process, SOP) for all sites. The company saved financial resources (as several systems were not maintained), compliance was improved, and it brought more clarity to all. Industry Quality leaders cannot do the same with respect to regulatory requirements, but if we speak with one voice, we can send a very clear signal that we expect regulators to establish one standardized science and data-driven global approach. As an example, at a global level, it makes no financial sense that the same PAC has many different national reporting requirements and that it requires many redundant reviews (one per country). At one company I worked for, 80 % of all CMC Regulatory Affairs resources were dedicated to managing PACs.
In addition to advocating for a gradation in regulatory oversight of pharma companies and a standardized global approach to regulatory requirements, the community of industry Quality leaders should be more active and lead other topics in the public health debate. One of these is how we – through better performance – can improve the perception of quality management, which FDA has stated they have ‘low confidence’ in. The majority of pharma companies demonstrate good quality performance and I’ve seen and met hundreds of Quality leaders that are doing an amazing job of ensuring safe, efficacious, quality products to patients. But there are some that do not. As a community of Quality leaders, we should voice our disapproval of Quality leaders and CEOs that do not operate with integrity causing drug shortages, poor perception of our community, and increasing reporting requirements for all. At the same time, we should offer our help to educate all senior pharma leaders and provide Quality leaders with support where that is needed. We can do more.
In summary, to reduce drug shortages, regulatory oversight of the pharma industry should be more differentiated with more control of non-compliant companies and more trust and flexibility for companies with good quality performance. The community of Quality leaders should be more vocal and advocate for a globally standardized approach to regulatory requirements for common topics, as well as work on improving the reputation of Quality management through integrity and good performance.